In Sponsored education
Learn from key healthcare brands with a range of sponsored education to help your understanding of how to use and advise on products in the pharmacy.Bookmark
SCROLL TO START LEARNING
STEP 1
UNDERSTANDING THE HAY FEVER STRUGGLE
During hay fever season, pharmacies support a wide range of customers, from those with mild symptoms to many struggling with more severe hay fever. However, pharmacy teams should always consider if customers are receiving the best treatment outcome, or if they are simply "putting up with symptoms." In particular, they should consider other factors like, the impact of polypharmacy and the customer's quality of life.
> THINK ABOUT THE FOLLOWING STATEMENTS and consider if they are TRUE or FALSE.
| TRUE | FALSE | |
APPROXIMATELY 2 OUT OF 3 PEOPLE WITH HAY FEVER HAVE MODERATE-SEVERE SYMPTOMS1 |
||
HAY FEVER SYMPTOMS SYMPTOMS, PARTICULARLY WHEN MODERATE-SEVERE, REDUCE QUALITY OF LIFE, INCLUDING DISRUPTION TO SLEEP, DAILY TASKS, OUTDOOR ACTIVITIES AND WORK2 |
||
MANY HAY FEVER SUFFERERS FREQUENTLY CONTINUE TO EXPERIENCE SEVERE SYMPTOMS WHILE TAKING TREATMENT3 |
||
ALMOST 3 OUT OF 4 HAY FEVER SUFFERERS USE MULTIPLE MEDICINES, OFTEN CONCURRENTLY3,4 |
||
MANY HAY FEVER SUFFERERS USE 3 OR MORE MEDICINES BEFORE SPEAKING TO A PHARMACIST5 |
As you can see, hay fever can have a significant impact on sufferers, and unfortunately climate change is making this worse.6 Rising temperatures are leading to longer pollen seasons and higher pollen levels, meaning customers may experience symptoms for longer.6 As many sufferers do not consult their pharmacist5 and instead self-select multiple treatments, pharmacy teams play a vital role in intervening to prevent polypharmacy and to ensure effective, appropriate treatment.
As you already know, there are various types of treatments available OTC for the management of hay fever symptoms, including antihistamines and nasal corticosteroids. Customers often resort to using multiple products, multiple times a day, which can be tiresome and sometimes ineffective.
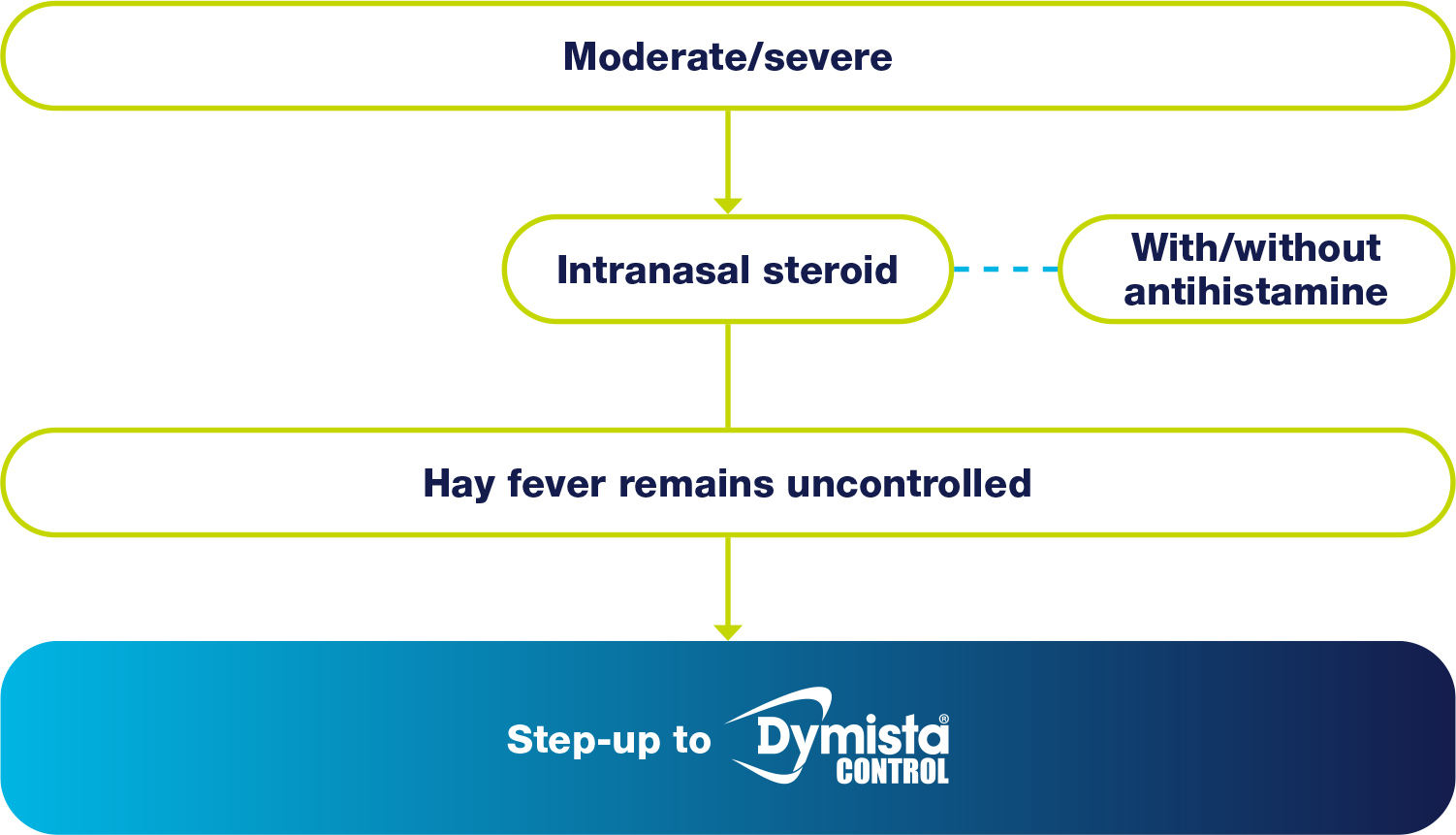
In particular, many customers suffering from moderate-severe symptoms (bothersome enough to reduce quality of life) may have tried an intranasal steroid spray (with or without oral antihistamines), but still suffer from troublesome symptoms.
FOR THESE CUSTOMERS, CONSIDER STEPPING UP TO DYMISTA® CONTROL.
WHEN HAY FEVER IS...
STEP 2
TREAT HAY FEVER WITH DYMISTA® CONTROL
Dymista® CONTROL can break the cycle of insufficient relief
Dymista CONTROL (P), Azelastine hydrochloride 137 micrograms / Fluticasone propionate 50 micrograms per actuation
ANTIHISTAMINES
Help alleviate symptoms like sneezing, runny nose and itching by blocking the affects of histamine.8 When used intranasally, the effects occur locally, providing quick relief. However, currently there are no nasal antihistamines available OTC.9
INTRANASAL CORTICOSTEROIDS
Reduce the inflammation in the nasal passages to treat nasal congestion, and various other symptoms of rhinitis.8
Dymista® CONTROL
Faster and more effective relief at reducing all EYE and NASAL hay fever symptoms than a steroid nasal spray alone (fluticasone propionate)3,10 or a steroid spray combined with an oral antihistamine(loratidine)11
-

Itchy, red,
watery eyes -

Sneezing
-

A runny nose &
nasal congestion
The British Society for Allergy & Clinical Immunology (BSACI) recommends:8
Treatment with a combined intranasal steroid and antihistamine is more effective than either agent used alone or than using a steroid nasal spray + oral antihistamine.
Discussing Dymista® CONTROL is as simple as...7
-
Assess hay fever symptoms
Are your symptoms bothersome, affecting your sleep or other aspects of daily life?
Yes
symptoms are bothersome; hay fever is moderate-to-severe
-
Query previous treatment
Have you used a steroid nasal spray for hay fever before?
Yes
symptoms are not sufficiently relieved with steroid sprays
-
Check for contraindications*
Are you pregnant or breastfeeding and/or using other medicines containing corticosteroids?
No
contraindications

*Refer to sections 4.3 and 4.4 of the SmPC.
STEP 3
USING DYMISTA® CONTROL CORRECTLY
Correct use of nasal sprays is crucial to achieve the desired effects. It is therefore very important to teach your customers how to use their spray correctly so that they have effective, fast-acting relief from their hay fever symptoms.
Advise your customer to use:7

1 spray per nostril, twice daily (morning and evening)
Correct technique can reduce the possibility of an unpleasant taste or smell1
Tilt the head forward slightly, as though reading a book
Breathe in lightly through the nose
If hay fever symptoms persist after 7 days of using Dymista® CONTROL, the patient should visit their GP to rule out an alternative diagnosis.7

1 bottle provides 1 month of relief for all eye and nose symptoms*
*RRP: £19.99 per bottle (120 sprays) 4 sprays a day: 67p a day.
People with hay fever can use Dymista® CONTROL for 3 months continuously. This should offer relief for their entire allergy season. For use beyond 3 months of continuous use, refer them to a GP.7
Dymista® CONTROL has an established safety profile and is well tolerated7,10

Correct technique can reduce the possibility of some side effects; however, there are other side effects that are mostly mild and typical of those reported with azelastine hydrochloride and fluticasone propionate, including:7
Nosebleeds: very common (≥1/10)
Headache: common (≥1/100 to <1/10)
Unpleasant taste or smell: common (<1/100 to <1/10)
Please consult the Dymista® CONTROL SmPC for full safety information.7
Dymista CONTROL (P), Azelastine hydrochloride 137 micrograms / Fluticasone propionate 50 micrograms per Actuation
SUMMARY
Help break the cycle of insufficient relief
THE STRUGGLE
RECOMMENDING
DYMISTA® CONTROL

Almost 3 in 4 hay fever sufferers use multiple medicines, often at the same time, and continue to experience severe symptoms.3,4

They become stuck in a cycle of insufficient relief.3,4
RECOMMENDING
DYMISTA® CONTROL
Faster and more effective at reducing all eye and nose symptoms than a steroid nasal spray alone (FP)7,10 or FP + oral antihistamine (LORA)7,11

*Recommended by The British Society for Allergy & Clinical Immunology (BSACI) as part of stepwise treatment approach8
FP=fluticasone propionate; LORA=loratadine; OTC=over-the-counter.

The only OTC 2-in-1 solution9
for adults with moderate-to-severe hay fever who have tried a steroid nasal spray7



